Text by Kristiina Mark
Plant Physiology Lab at our university hosts many inspiring and dedicated female researchers working in the field of plant sciences. Here we cheer on our feminine brain power, that currently includes researchers and associated professors Dr. Astrid Kännaste, Dr. Assoc. Prof. Eve Runno-Paurson, Dr. Inga Jüriado, Dr. Kaia Kask, Dr. Kristiina Mark, Dr. Assoc. Prof. Tiina Tosens, Dr. Triinu Remmel and junior researchers Congmiao Xie, Helina Nassar, Jesamine Jöneva Rikisahedew, Upasana Sharma, Vivian Kuusk and support stuff MSc. Evi Vaino, MSc. Piret Pärlist, MSc. Mari Tobias, MSc. Tiia Kurvits.
To emphasize the capacity of our collegium, lets run some simple statistics.
- This group of colleagues includes project managers, lecturers/teaching professors, research project leaders, lab managers, technicians/assistants, supervisors, PhD students.
- They have technical and scientific capacity in wide range of plant science fields, such as plant anatomy, plant stress, biogenic volatile organic compound (VOC) measurements, measurements of gas-exchange and photosynthetic parameters, plant pathogen research, lichenology, symbiosis biology, plant-insect interactions, implementation of novel agricultural crops, plant biochemistry, etc.
- As combined, they have research work experience and competence from working in at least 9 different countries (Australia, China, Estonia, Finland, India, South Africa, Sweden, Switzerland, USA).
- Among our women scientists are principal investigators of 5 currently running funded research projects, while many are also participating as senior research stuff in 5 currently funded research projects. In their total career they have carried out (at least) 63 funded research projects (out of which 17 as the principal investigators).
- In total we have dedicated 179 years to work in science (calculated as sum of years starting from PhD studies) and have published at least 173 research articles (indexed as 1.1., as indicated in the Estonian Science Info System, by 07.02.2024).
But what inspires the women in our Plant Physiology Lab?

“I am currently conducting research in the field of plant ecophysiology where I am hoping to draw links between anatomy and physiology. Being at the forefront of research allows me to grow by being challenged daily, and I am inspired by other successful women who have found their voices in science. My favourite part of what I do is getting to conduct experiments in the lab.”
-Jesamine Rikisahedew, PhD student and junior researcher

“Research fields are plant physiology and ecophysiology. I am interested in the inner workings of the cornerstone process of life on Earth, photosynthesis, and how it has adapted to various environments, as well as how to improve it for the sake of climate-resilient crops.”
– Dr. Tiina Tosens, Associate professor

„Olen põllumajandusteadlane, kes huvitub põllukultuuridest ja nende tervisest ning seda juba ligi 23 aastat. Liigun põllul ja laboris. Viimastel aastatel on lisandunud Eestis uute põllukultuuride kasvatamise uurimine ja populariseerimine. Eriline armastus on kartuli ja bataadi vastu.“
– Dr. Eve Runno-Paurson, Associate professor

“I work on the symbiotic interactions of lichen, and the diversity and evolution of lichen secondary metabolites for my thesis. The beauty and diversity of lichen is brightly shining in my world all the time when I came across lichen during my master study.“
– Congmiao Xie, PhD student and junior researcher

“I conduct research mostly on cryptogam biology, ecophysiology and ecology. This includes mosses, lichens and algae. Many things about these organism groups are still unknown, and it is absolutely fascinating to study the physiology (aka “behaviour”) of someone you might consider as an alien (and it’s not a joke – some of them easily survive outer space and Mars conditions!). My work is my inspiration, the daily challenges and thrilling discoveries, but I also highly appreciate inspiring interactions with students, their personal, academic and scientific growth, and thriving collaboration with colleagues.”
– Dr. Kristiina Mark, researcher

“My research field is plant stress and volatiles. My inspiration is constant stress :D”
– Dr. Kaia Kask, researcher

“Olen nooremteadur ja aednik. Mulle meeldivad taimed ja nende heaolu eest hoolt kanda nii tubastes tingimustes kui põllul. Teaduses huvitab kartul ja tema kahjustajad.“
– Helina Nassar, PhD student and junior researcher
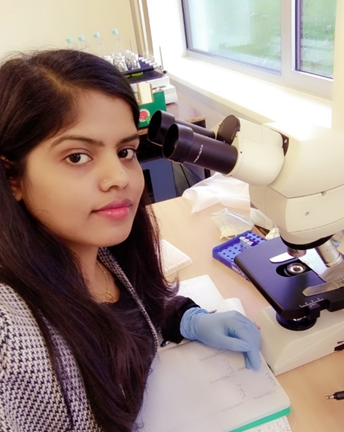
“My field of research is rooted in ecophysiological and anatomical studies of cryptogams; mostly lichens and mosses, where I aim to determine how these organisms acclimate to changes in various environmental conditions. My research consists of fieldwork and lab work, where I get to experience a balance of both worlds. I greatly enjoy visiting the natural habitats (forests and natural parks) of the organisms I study where I can tune in to nature. My inspiration is my idols in the research field, and every woman who adds value to their fields of science, which pushes me to be motivated and upscale my skills in research as well as in life.”
-Upasana Sharma, PhD student and junior researcher





















